Microbiology
The QIAGEN® QIAstat-Dx® Respiratory SARS-CoV-2 Panel

This multiplex syndromic cartridge detects and differentiates between 22 respiratory targets from nasopharyngeal swabs (NPS) eluted in universal transport media (UTM). The QIAstat-Dx Respiratory SARS-CoV-2 Panel is intended for the detection and differentiation of nucleic acid from SARS-CoV-2 and the following organism types and subtypes: Adenovirus, Coronavirus 229E, Coronavirus HKU1, Coronavirus NL63, Coronavirus OC43, SARS-CoV-2, Human Metapneumovirus A+B, Influenza A, Influenza A H1, Influenza A H3, Influenza A H1N1/pdm09, Influenza B, Parainfluenza virus 1, Parainfluenza virus 2, Parainfluenza virus 3, Parainfluenza virus 4, Rhinovirus/Enterovirus, Respiratory Syncytial Virus A+B, Bordetella pertussis, Chlamydophila pneumoniae, and Mycoplasma pneumoniae.
Features:
– Detection of 22 respiratory targets in about an hour
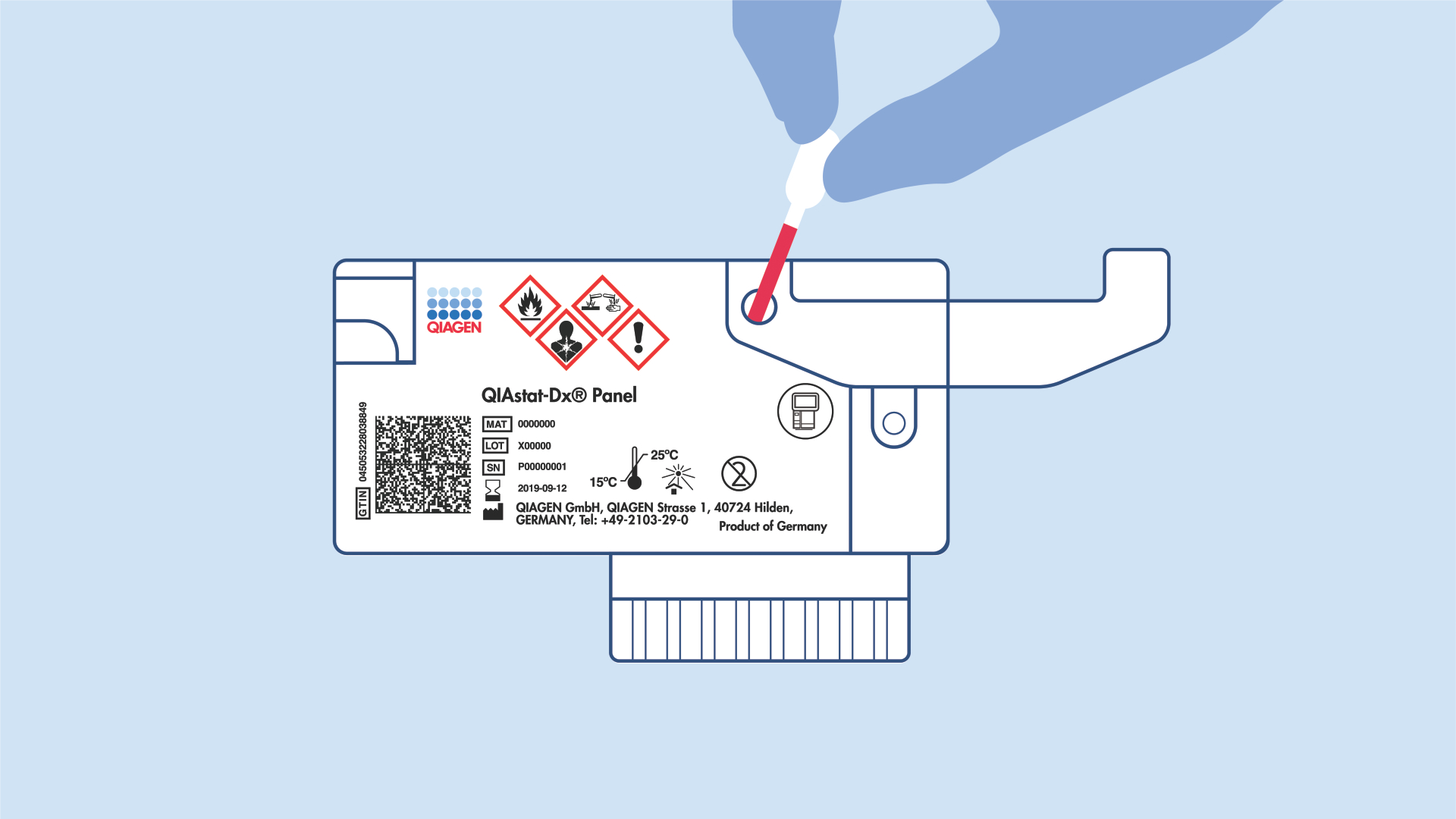
– Intuitive workflow with less than one-minute hands-on time
– No precision pipetting required
– Wet and dry reagents are preloaded
– Room temperature stable
– 6 test cartridges and transfer pipettes included in each kit